EMT2 Clinical Trial

OVERVIEW
Recruitment to the study has now closed. The patients will take the capsules for the next 2 years so the results will be due in 2026.
This clinical trial has been designed to test whether a naturally-occurring omega-3 fatty acid called eicosapentaenoic acid (EPA for short) improves outcomes in patients who have an operation for the removal of bowel (also known as colorectal) cancer that has spread to the liver.
A small trial in Leeds suggested that a high-purity, high-dose preparation of EPA might be beneficial but only a bigger trial can give us a definite answer as to whether taking EPA will work. The EPA capsules are being compared with dummy (placebo) capsules so that we can be sure that EPA is or isn’t better than no supplement at all.
Taking part involves starting trial capsules before the operation and then continuing them after surgery even if the bowel cancer comes back. There are trial visits every 6 months that can either be combined with outpatient clinic visits or by telephone, if the hospital is a long way away.
Trial tests include the check-up CT scans that would be performed by the hospital department anyway, but also questionnaires about quality of life and some patients will be asked for extra blood tests.
Participant Information Downloads
NEWS
STUDY TIMELINE
Recruitment to the study has now closed. The patients will take the capsules for the next 2 years so the results will be due in 2026.








PARTNERS
The trial has been approved by a Research Ethics Committee, Health Research Authority and the Medicines & Healthcare Regulatory Authority who oversee all drug trials in the UK
FREQUENTLY ASKED QUESTIONS
 NOW THAT I HAVE FINISHED THE TRIAL CAPSULES TREATMENT, IS THERE AN ALTERNATIVE THAT I CAN TAKE?
NOW THAT I HAVE FINISHED THE TRIAL CAPSULES TREATMENT, IS THERE AN ALTERNATIVE THAT I CAN TAKE?
Until the EMT2 trial results are evaluated and published, there is no evidence that taking EPA is beneficial to patients who have had surgery for bowel cancer spread to liver. In the EMT2 trial, patients took a specific, high-dose form of EPA and therefore the study results (beneficial or otherwise) cannot be assumed to be similar for other omega-3 preparations with a different dose or level of purity.
By way of comparison, the daily dose of pure EPA in the trial was 4 x 1000 milligram capsules compared with typical omega-3 capsules available in Health Food outlets, which usually contain 200 milligrams EPA per capsule, and which include a mixture of other substances.
Overall, it is not recommended to take an alternative EPA or other omega-3 preparation to replace the trial treatment until the EMT2 trial results are available.
As soon as we can, we will describe the results of the trial in Plain English to you and family members, if you wish to know.
 WILL MY CANCER TREATMENT BE ALTERED BY JOINING THE TRIAL?
WILL MY CANCER TREATMENT BE ALTERED BY JOINING THE TRIAL?
Your standard of treatment and care will not be affected by either volunteering or not taking part in the trial. Always remember that you can withdraw from the trial at any point, without giving a reason.
 WHAT IS EPA?
WHAT IS EPA?
EPA is one of the main omega-3 fatty acids found naturally in oily fish like sardines and mackerel, along with another fatty acid called DHA. DHA is thought to be important for brain and nerve development and function, whereas EPA has anti-inflammatory effects. The best evidence that omega-3s have anti-cancer activity is for EPA, which is why we are testing EPA on its own in EMT2.
 WHAT ARE THE CAPSULES BEING USED IN EMT2?
WHAT ARE THE CAPSULES BEING USED IN EMT2?
The EPA capsules in EMT2 are pure EPA and give a high dose of EPA. The same capsules and dose of EPA has recently been shown to reduce risk of heart attack and stroke in individuals at high risk of such events in an international trial. The capsules (EPA and placebo capsules) are provided by Amarin Corp. and are called Vascepa®, which is already licensed for use in people with high blood lipid levels in the USA. The EPA is derived from fish and the capsule contains gelatin from an animal source so the trial is not suitable for Vegetarians.
 WHY NOT TAKE AN OMEGA-3 CAPSULE AVAILABLE FROM THE SHOPS OR ONLINE?
WHY NOT TAKE AN OMEGA-3 CAPSULE AVAILABLE FROM THE SHOPS OR ONLINE?
Omega-3 capsules available on the high street contain a much lower dose of EPA than the trial capsules (approximately three times less EPA than a Vascepa® capsule). The fatty acid content in high street capsules is a complex mix of the multiple fatty acids found in fish oil, including some omega-6 fatty acids, the effects on cancer of which are unknown. We stipulate that no other omega-3 fatty acid supplements should be taken during the trial.
 DOES EPA CAUSE ANY SIDE-EFFECTS?
DOES EPA CAUSE ANY SIDE-EFFECTS?
Some people may experience some mild side effects relating to their digestive system such as nausea and diarrhoea, but these often can be minimised if the capsules are taken with meals.
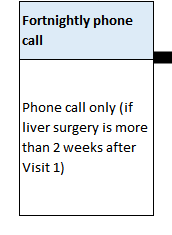
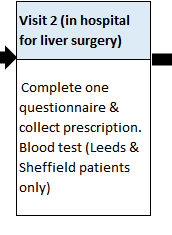
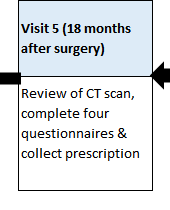
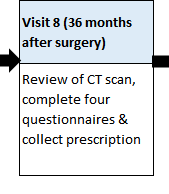
 HOW MANY TRIAL VISITS ARE THERE?
HOW MANY TRIAL VISITS ARE THERE?
There will be regular contact by either telephone or study appointments (maybe 5-10), which will link to your standard care appointments, wherever possible. If transport is a problem then telephone follow-up can be done and capsules posted by Courier.
These visits will include a review of your progress, a health questionnaire, a prescription for new capsules and, for some participants, a blood test.
 IS ALL MY TRIAL INFORMATION CONFIDENTIAL?
IS ALL MY TRIAL INFORMATION CONFIDENTIAL?
Confidentiality during the trial is important, and the information collected about you will be handled in accordance with the consent form and the 1998 Data Protection Act.
RECRUITMENT
Patients Recruited
Recruitment closed on 30/11/2023.
RECRUITING CENTRES
| Basingstoke & North Hampshire Hospital | Basingstoke |
| Churchill Hospital | Oxford |
| Queen Elizabeth Hospital | Birmingham |
| St James’s Hospital | Leeds |
| Queens Medical Centre | Nottingham |
| Northern General Hospital | Sheffield |
| Southampton General Hospital | Southampton |
| University Hospital Aintree | Liverpool |
| Freeman Hospital | Newcastle |
| Royal Free Hospital | London |
| Addenbrookes Hospital | Cambridge |
| King's College Hospital | London |
| University Hospital of Wales Hospital | Cardiff |
TRIAL TEAM




